Moderna to ask Health Canada to authorize its COVID-19 vaccine for children aged six months to five years old.
Moderna says it's working on a submission to Health Canada for the approval of a COVID-19 vaccine for children under the age of 6.
Earlier today, the company asked regulators in the U.S. to authorize low doses of its vaccine for kids aged six months to five years old.
So far, Health Canada hasn't approved any COVID-19 vaccines for kids younger than 5.
To date, the Pfizer Comirnaty vaccine has been approved for kids aged five to 11, and Moderna's Spikevax shot has been authorized for children aged six to 11.



 Oxford OPP Lay Charges After Warrant Execution
Oxford OPP Lay Charges After Warrant Execution
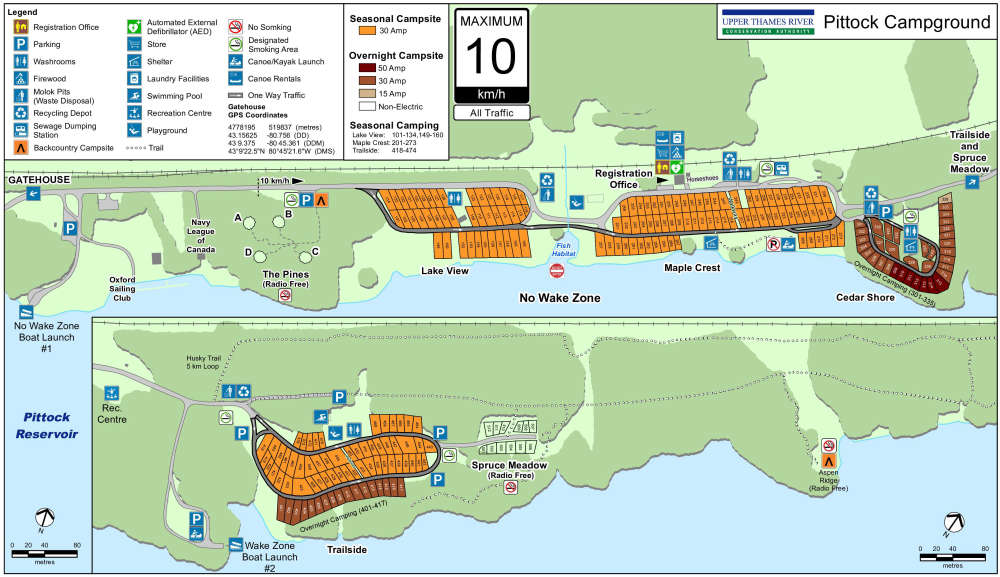 Camping Season is Open at Pittock
Camping Season is Open at Pittock
 Application Window Open for Oxford County Grants Program
Application Window Open for Oxford County Grants Program
 PJHL Preview - Schmalz Cup Final
PJHL Preview - Schmalz Cup Final
 Oxford Winds Celebrates 30 Years
Oxford Winds Celebrates 30 Years
 Nomination Period Opens for Municipal Election
Nomination Period Opens for Municipal Election
 Theft in Thamesford is Under Investigation
Theft in Thamesford is Under Investigation
 Winning Lotto 6/49 Ticket Sold in Oxford
Winning Lotto 6/49 Ticket Sold in Oxford
 Thamesford Lions Club Celebrates 65 Years
Thamesford Lions Club Celebrates 65 Years
 Yellow Frost Advisory Issued for Oxford
Yellow Frost Advisory Issued for Oxford
 News Poll: Schmalz Cup 2026
News Poll: Schmalz Cup 2026
 Realtors Care Food Drive Returns Next Week
Realtors Care Food Drive Returns Next Week
 PJHL Playoff Recap - April 29th
PJHL Playoff Recap - April 29th
 Charges Laid After Altercation in Woodstock
Charges Laid After Altercation in Woodstock
 New Partnership Protects Land in Oxford
New Partnership Protects Land in Oxford
 Bomb Threat Unfounded in Woodstock
Bomb Threat Unfounded in Woodstock
 Big Projects Announced for TVDSB
Big Projects Announced for TVDSB
 Tick Season Reminders from Southwestern Public Health
Tick Season Reminders from Southwestern Public Health
 PJHL Playoff Recap - April 28th
PJHL Playoff Recap - April 28th
 Highway Construction Approaching in Burgessville
Highway Construction Approaching in Burgessville



Comments
Add a comment