Moderna to ask Health Canada to authorize its COVID-19 vaccine for children aged six months to five years old.
Moderna says it's working on a submission to Health Canada for the approval of a COVID-19 vaccine for children under the age of 6.
Earlier today, the company asked regulators in the U.S. to authorize low doses of its vaccine for kids aged six months to five years old.
So far, Health Canada hasn't approved any COVID-19 vaccines for kids younger than 5.
To date, the Pfizer Comirnaty vaccine has been approved for kids aged five to 11, and Moderna's Spikevax shot has been authorized for children aged six to 11.



 Woodstonian Charged in Child Exploitation Case
Woodstonian Charged in Child Exploitation Case
 Woodstock Home and Lifestyle Show Returns this Weekend
Woodstock Home and Lifestyle Show Returns this Weekend
 Nominations Open for Tillsonburg Citizen of the Year
Nominations Open for Tillsonburg Citizen of the Year
 PJHL Playoff Recap - March 17th
PJHL Playoff Recap - March 17th
 UPDATE: Video Footage Wanted for Death Investigation
UPDATE: Video Footage Wanted for Death Investigation
 MURC: Coming Soon to Ingersoll!
MURC: Coming Soon to Ingersoll!
 Candidate Information Session Coming Up
Candidate Information Session Coming Up
 EZT Remembers Former Ward 3 Councillor
EZT Remembers Former Ward 3 Councillor
 SWPH Issues Cold Weather Alert
SWPH Issues Cold Weather Alert
 UPDATE: Strong Winds Continue this Morning
UPDATE: Strong Winds Continue this Morning
 Woodstock Resident Charged with Speeding
Woodstock Resident Charged with Speeding
 Interview with the Mayor - March 16th, 2026
Interview with the Mayor - March 16th, 2026
 Woodstock Driver Charged in Brant County
Woodstock Driver Charged in Brant County
 PJHL Playoff Recap - March 13th to March 15th
PJHL Playoff Recap - March 13th to March 15th
 March Break Safety Tips from Oxford OPP
March Break Safety Tips from Oxford OPP
 Oxford-Elgin Rainbow Research Project Underway
Oxford-Elgin Rainbow Research Project Underway
 OPP: Plan Ahead Before St. Paddy's Day Festivities
OPP: Plan Ahead Before St. Paddy's Day Festivities
 Another Friday the 13th in Port Dover Today
Another Friday the 13th in Port Dover Today
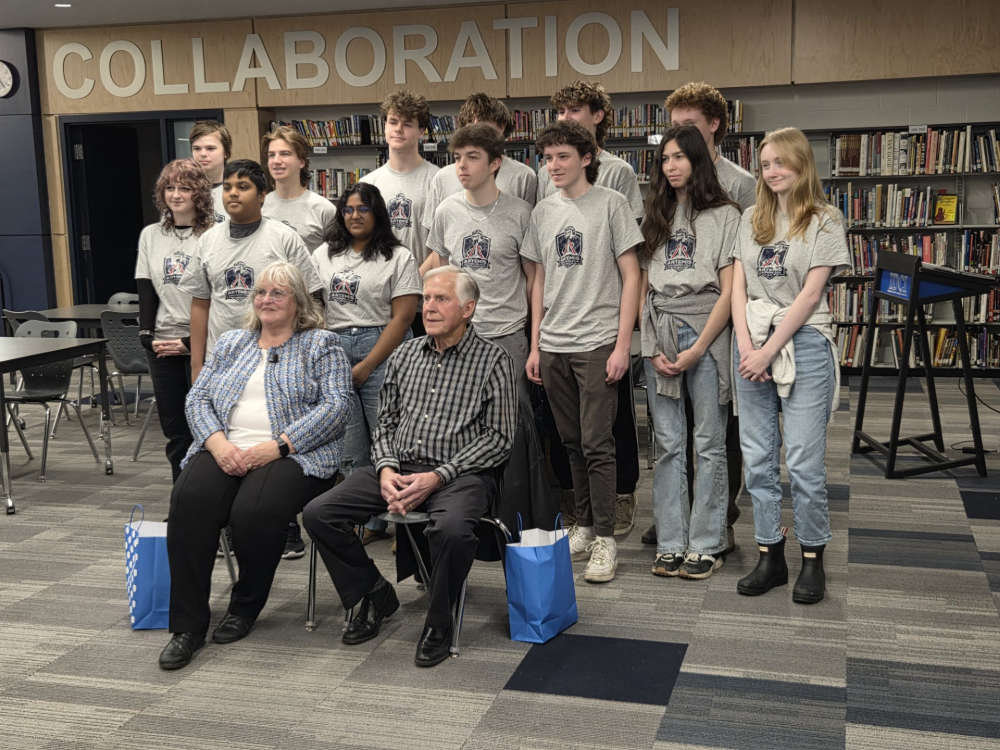 Hansen Parents Visit Artemis II Explorers Club at IDCI
Hansen Parents Visit Artemis II Explorers Club at IDCI
 East Oxford Parents Respond to AARC Announcement
East Oxford Parents Respond to AARC Announcement



Comments
Add a comment